This April, Lisa Peter, an 11-year-old student at Kambange Primary School in Dar Es Salaam, was sprawled on the living room couch after school, scrolling through television channels, when an advert by the Ministry of Health on national television grabbed her attention.
It showed a family doting on a young girl, getting her ready for school. But overlaid on the familiar scene was a message about a huge vaccination campaign against cervical cancer – a disease that kills over 7,000 women each year in Tanzania. The advert cut to smiling girls playing at school. Lisa felt jolted, recognising herself in the advert’s message.
“The teacher told me – this little pinch means a huge difference! Now I’m protected against cervical cancer.”
– Lisa Peter, age 11, student at Kambange Primary School in Dar Es Salaam
“I told my mother I also wanted protection from cancer,” Lisa says.
Lisa is one of the 5 million Tanzanian girls aged 9-14 years who were targeted in a recent vaccination campaign against the human papillomavirus (HPV), the virus that causes the vast majority of cervical cancers, widely known in Tanzania as ‘saratani ya shingo ya kizazi’.
Kicking off in the third week of April as countries around the world observed World Immunization Week, the campaign was notable not just for its scale, but for its approach. Fuelled by research from Tanzania’s own scientists, who had been investigating the effectiveness of a single-dose HPV vaccine, the campaign inaugurated a shift from the two-dose regimen used previously.
For the country’s health services, that meant lowered logistical barriers, financial savings, the loosening of supply constraints. For kids like Lisa, it meant lifelong protection would come at the cost of just a single poke.
Unlike parents swayed by vaccine rumours that can hold back the vaccination efforts, Lisa got instant reassurance from her mother who – like many parents in her area – had been sensitised by a community health worker (CHW) in the city’s Kinondoni Municipality about the vaccine’s safety and health benefits.
Soon, at school, it was Lisa’s turn to get immunised. “It hurt my shoulder a bit,” Lisa told VaccinesWork shortly afterwards. “The teacher told me – this little pinch means a huge difference! Now I’m protected against cervical cancer,” she said.
Marygoreth Temba, Lisa’s schoolteacher and the person responsible for health at Kambange Primary School, says she collaborated with health workers deployed by the Ministry of Health at schools, and helped in dispelling fears and breaking down myths surrounding vaccines.
“As an educator, I made sure pupils got accurate messages. This seems to have trickled down to parents as well. I’ve seen many parents change their attitudes and accept the vaccines.”
– Marygoreth Temba, schoolteacher
“As an educator, I made sure pupils got accurate messages. This seems to have trickled down to parents as well. I’ve seen many parents change their attitudes and accept the vaccines,” said Temba, echoing the voices of those who were on the frontline across Tanzania, advocating and providing vaccination during the campaign from 22 to 26 April, with support from Gavi, the Vaccine Alliance, UNICEF, and the World Health Organization (WHO).
While many countries have implemented HPV vaccination programmes over the past decade, Tanzania’s approach has been unique. Launched in 2019, the vaccine was immediately offered as part of the routine immunisation schedule for 14-year-olds . At that time, Tanzania had the fourth highest incidence of cervical cancer globally, with 59 cases per 100,000 women, underscoring the urgency of the vaccine’s roll-out. The incidence is higher in countries like Tanzania due to high HIV/AIDS rates, which places women at a higher risk of getting cervical cancer.
With the increase of global supply, the vaccine can now be offered to a larger target group, spanning the ages nine to 14, in a nationwide campaign.
“Vaccinating young girls before they are exposed to HPV is crucial,” says Dr Norman Jonas, a tutor in Internal Medicine at the Kilimanjaro Christian Medical College (KCM College). Jonas has participated in public awareness campaigns about HPV vaccines as part of the health promotion support team appointed by the Ministry of Health. He says, “The earlier we protect them, the lower their risk of developing cervical cancer later in life.” This is particularly vital in contexts like Tanzania’s, where cervical cancer screening services are still limited in availability.
Routine vaccination was disrupted by the COVID-19 pandemic. Schools – the main site for HPV vaccination – closed, and opportunities to protect girls were missed.
April’s multi-age cohort (MAC) campaign – which comes five years after the vaccine’s launch in Tanzania – allowed the country’s immunisation system an opportunity to reach all girls aged 9–14 years, an age-range calculated to optimise the vaccine’s impact with a single dose of the cancer-blocking vaccine
Clinical trials on the effectiveness of a single dose had been ongoing in the country, spearheaded by the National Institute for Medical Research (NIMR).
John Changalucha, is a Senior Research Scientist at NIMR who is part of the clinical trials whose findings were presented to WHO and the Ministry of Health, leading to a switch to single-dose vaccine. He says the shift to a single dose was further spurred by past experience with “logistical challenges, and the higher costs associated with offering multiple doses” .
97% reached in just one week
“The campaign response was positive and successful,” said Dr Florian Tinuga, Program Manager of the Immunization and Vaccination (IVD) Program of the Ministry of Health.
“Vaccinating young girls before they are exposed to HPV is crucial. The earlier we protect them, the lower their risk of developing cervical cancer later in life.”
– Dr Norman Jonas, Kilimanjaro Christian Medical College
According to field activity reports from the Ministry of Health, the campaign reached an impressive 97% of the targeted 5 million girls by the end of the week of the campaign in April. Over 87% were from schools, with the remaining girls reached through non-school vaccination centres.
CHWs like Chuma Bakari played a critical role in dispelling myths and encouraging participation in the HPV vaccination campaign. Bakari, who works in Tanzania’s Kinondoni Municipality in Dar es Salaam, explains one of the challenges he encountered: “Some parents expressed concern about the age at which the vaccine was being administered.”
To address these concerns, Bakari intensified his community outreach efforts. “As part of my routine health education activities, I focused specifically on cervical cancer and the HPV vaccine during the campaign,” he says. “I went from door to door to provide accurate information, and I’m happy to say I’ve witnessed a positive shift in attitudes. In fact, some parents even brought their younger daughters, who weren’t yet eligible, hoping to get them vaccinated.”
“I went from door to door to provide accurate information, and I’m happy to say I’ve witnessed a positive shift in attitudes. In fact, some parents even brought their younger daughters, who weren’t yet eligible, hoping to get them vaccinated.”
– Chuma Bakari, community health worker, Kinondoni Municipality
The country is now leveraging the lessons learnt from the campaign as it embarks on a sustainable immunisation plan that runs until December this year, aimed at ensuring all girls aged 9 to 14 receive the vaccine.
The IVD Program Manager, Dr Tinuga, says, “We will revisit hard-to-reach areas where girls might have missed opportunities. All those who missed the vaccine will be reached through routine immunisation programmes and periodic intensification of routine immunisation.”
Source : https://www.gavi.org/vaccineswork/tanzanias-5-million-girls-hpv-vaccination-campaign-success
Sexually transmitted infections (STIs), such as chlamydia, syphilis, and gonorrhea, have been on the rise nationwide. They disproportionally affect men who have sex with men and transgender women. Left untreated, they can lead to serious health issues, such as blindness, brain and nerve problems, and infertility in women. Condoms can block many STIs. However, condoms are not always used consistently or correctly. So researchers have been exploring other options for preventing STIs, especially among those at elevated risk for repeated infections.
Previous studies found evidence that the antibiotic doxycycline, taken shortly after sex, might reduce the risk of bacterial STIs among men who have sex with men. This approach is called doxycycline postexposure prophylaxis, or doxy-PEP. However, some experts have been concerned that preventive use of antibiotics could lead to antibiotic resistance. This might reduce future options for treating STIs and other bacterial infections.
To learn more, a team led by scientists at the University of California, San Francisco (UCSF) and the University of Washington, Seattle, set out to measure the safety and effectiveness of doxy-PEP. They also looked for evidence of antibiotic resistance. Results were published in the New England Journal of Medicine on April 6, 2023.
The study enrolled 501 adults considered at high risk for bacterial STIs, either men who had sex with men or transgender women. All had been diagnosed with a bacterial STI in the past year and reported having sex without using a condom in the past year. They were either living with HIV or were taking or planning to take medication to prevent HIV infection.
Participants were randomly assigned to receive either doxy-PEP or standard care. Those in the doxy-PEP group were told to take a 200 mg doxycycline tablet as soon as possible within 72 hours after condomless sex. Participants were tested for STIs every three months and followed for one year.
The researchers found that the doxy-PEP group had a two-thirds lower incidence of syphilis, gonorrhea, and chlamydia compared to the standard-care group during each three-month time period. STIs were detected in about 10% of the quarterly tests administered to those in the doxy-PEP group, compared to about 30% of those in the standard care group.
Gonorrhea was the most often diagnosed STI. The incidence of gonorrhea per quarter in the doxy-PEP group was about 55% lower than in the standard care group. Chlamydia and syphilis were each reduced by more than 80% per quarter.
The researchers found that the doxy-PEP group had a modestly higher proportion of doxycycline-resistant Staphylococcus aureus living in the nose after 12 months. The incidence of gonorrhea strains resistant to the antibiotic tetracycline, which is in the same antibiotic class as doxycycline, was 38.5% in the doxy-PEP group compared to 12.5% in the group with standard care. This finding suggests doxy-PEP could be less effective in preventing gonorrhea with existing tetracycline resistance; however, the number of available gonorrhea cultures was low.
“It will be important to monitor the impact of doxy-PEP on antimicrobial resistance patterns over time, and weigh this against the demonstrated benefit of reduced STIs and associated decreased antibiotic use for STI treatment in men at elevated risk for recurrent STIs,” says Dr. Annie Luetkemeyer of UCSF, a co-leader of the study. “Given its demonstrated efficacy in several trials, doxy-PEP should be considered as part of a sexual health package for men who have sex with men and transwomen if they have an increased risk of STIs.”
SOURCE:
https://www.nih.gov/news-events/nih-research-matters/antibiotic-can-help-prevent-common-sexually-transmitted-infections

Jacqueline Materu, a fellow in the Mwanza Fogarty Research Training Program, won the Best Paper Award at the 5th International Conference on Statistics: Theory and Applications (ICSTA 2023), which took place at Brunel University, London. This award was for the presentation of her paper titled “Comparison of survival analysis approaches to modelling age at first sex among youth in Kisesa, Tanzania.”
The ICSTA Best Paper Award is a competitive prize given to authors of the papers that rank highest in a peer review process by the esteemed session chairs of the ICSTA. As a Best Paper Award winner, Jacqueline received a certificate and a 25% discount toward registration for the 2024 ICSTA conference series. Moreover, Jacqueline can submit her modified presented paper to a related journal published by Avestia Publishing for free publication.The Mwanza Fogarty Research Training Program supported Jacqueline’s attendance at the 5th ICSTA. Read more about Jacqueline’s work and the Mwanza Fogarty Research Training Program here.
Cervical cancer is often called “a silent killer” because there are no obvious early warning signs. As the fourth most common cancer in women, it kills more than 300,000 a year globally.
But what if we could better protect against the virus that causes most cervical tumors?
The human papillomavirus, better known as HPV, is one of the most common sexually transmitted infections in the world because it often circulates without causing any disruptive symptoms. But it can cause genital warts and various cancers in both men and women—and is driving the recent rise of throat cancer in men.
Even though vaccines that prevent HPV have existed since 2006, experts say a new development may put the global goal of vaccinating 90 percent of girls under 15 within reach.
(STDs are at a shocking high. How do we reverse the trend?)
The revelation in question is simpler than you might expect: Emerging evidence suggests the HPV vaccines are effective after just one dose—rather than the two or three that are currently recommended in most countries. In 2022, the World Health Organization advised switching to a single dose of the vaccine, explaining that not only is it efficacious but it will also make distribution easier, particularly in low- and middle-income countries.
“These are remarkable vaccines. They’re incredibly potent in preventing infection,” says Andrew Pollard, chair the United Kingdom’s Joint Committee on Vaccination and Immunization. “I think we will see a world where cervical cancer is a rarity.”
The U.K is among more than 20 countries that have announced plans to switch to a single dose, a group that also includes Australia, Ireland, and Mexico. But will other countries follow suit?
A surprising discovery
The first hint that one dose of the vaccine works as well as two or three came somewhat unexpectedly—a result of studies in Costa Rica and India that were both testing multiple doses. In both cases, researchers later examined women who had only received a single dose—and those studies revealed it was still remarkably effective.
“There’s a very strong biological rationale for why we expect a single dose to work,” says Ruanne Barnabas, chief of infectious diseases at Massachusetts General Hospital who is leading the KEN SHE study, a randomized clinical trial investigating the efficacy of the single-dose vaccine in Kenyan women ages 15 to 20.
Typically, she explains, vaccines like the HPV vaccine are given in multiple doses because they only use part of the virus to elicit an immune response. But the proteins in the HPV vaccine behave a little differently: they reassemble themselves to mimic the whole virus—making it plausible that the vaccine could be more protective.
(How mRNA vaccines might help us fight other hard-to-treat cancers.)
More evidence has trickled in to support that hypothesis. Last year, the KEN SHE study reported that the single-dose had a 98 percent efficacy at 18 months—and new data released last month shows that has held strong through 36 months. Meanwhile, results from a study comparing vaccine efficacy in the Kenyan women to that of girls ages 9 to 14 in Tanzania showed that their immune responses were on par—suggesting a single dose could be just as effective in the younger age group.
Finally, follow-up studies of the women and girls from the original India and Costa Rica studies show that their immune responses continue to be strong more than a decade after accidentally getting just a single dose. To Pollard, this was a particularly compelling piece of evidence while weighing whether to recommend a change in the U.K’s vaccination program.
“If your antibody levels stay absolutely rock solid for 10 years, they’re not going to suddenly fall off a cliff,” he says. “The immune system doesn’t do that.”
Why does it matter?
If the evidence continues to build in favor of the single-dose vaccine, it would be a game changer—particularly in low- and middle-income countries.
These countries face major challenges in delivering HPV vaccines—and as a result they suffer the highest burden, or about 90 percent of cervical cancer deaths.
The first challenge is finding the people who need the vaccines in the first place, says Greg Widmyer, senior adviser for immunization at the Bill & Melinda Gates Foundation, which funded the KEN SHE study. It’s hard enough getting preteen and teenage girls into a clinic once for a shot but to come back for a second dose—or a third? “We were seeing a big drop,” he says.
Delivering multiple shots is also expensive —and not just for the jabs themselves. Widmyer points out that every time you administer a dose, you must pay for staff time, transportation, and other overhead costs. For some countries, these costs for a two- or three-dose vaccine can be so prohibitive that they don’t even bother to set up a vaccination program—other countries are just beginning to introduce the vaccine.
With the single dose, Greg says, the channel to reach girls “opens up completely”—you can set up vaccination clinics at places where girls already are, from schools to hair salons or other places in their communities.
(Why vaccines are critical to keeping diseases at bay.)
But it could also be a win everywhere. Switching to a single dose would ensure there’s more vaccine to go around—meaning countries would be able to vaccinate a broader range of people to reach herd immunity, or the point when enough people have been vaccinated or infected naturally that the population as a whole becomes immune to a disease. Barnabas points out that countries that already allow for vaccination among boys and older age groups have seen “a really dramatic” decrease in the number of precancerous lesions and warts and cancers.
For the U.K., Pollard says it was primarily a question of efficiency. “This decision really was driven by the emerging data, which made it very clear that we were not going to be losing protection by going down to one dose—and therefore, why wouldn’t you?”
An end to cervical cancer?
It’s not yet clear when—or whether—other countries will make the leap to just a single-dose of the vaccine. Some may wait for more evidence from studies that are ongoing, including a trial evaluating the efficacy of a single dose among 20,000 girls ages 12 to 16 in Costa Rica. That study is expected to reach its endpoint in two years.
But experts believe that momentum is building. “I firmly believe the elimination of cervical cancer is possible,” WHO Assistant Director-General Dr Princess Nothemba (Nono) Simelela said in a press release announcing the organization’s decision to advise switching to a single dose. “This single-dose recommendation has the potential to take us faster to our goal of having 90 percent of girls vaccinated by the age of 15 by 2030.”
Yet Pollard says it’ll likely be decades before we see the true impact on cervical cancer because so many women have already been infected. “What we’re doing is buying lives today that would have been lost in the future,” he says.
But he and other experts agree that it would be transformative—and provides more equity in the global healthcare system.
“In Africa, every family knows a woman who has suffered from this cancer. And this is an ugly cancer. It’s one that leads to a painful and somewhat dehumanizing end for these women,” Widmyer says. For those living in higher-income countries, he adds, “the privilege of not knowing somebody who has died this way is pretty unique.”
Source : https://www.nationalgeographic.com/science/article/cervical-cancer-hpv-vaccine-single-dose
The Mwanza Intervention Trial Unit (MITU) is a collaborative research Unit based at the National Institute for Medical Research (NIMR) campus in Mwanza, Tanzania. The mission of MITU is to contribute to improving health through the development and evaluation of interventions against HIV and other health problems by conducting research, including clinical trials to the highest international standards; to enhance the capacity to carry out such research in Tanzania, and the East African Region; and to contribute to the translation of research findings into health policy.
MITU is currently running several research projects in the Lake Zone including Mwanza, Geita, Shinyanga and Simiyu regions. The implementation of the ongoing research projects involves data collection from study participants residing in these regions and MITU projects’ staff members usually stay in the field in rural areas for several weeks. Due to high demand of vehicles to cater for current project activities, we are inviting interested companies and individuals to submit their bid to provide vehicle and driver services for the duration of 6 months.
Bids for supply of cars
We are inviting bids for supply of cars as specified below:
- The bidder to provide a vehicle and a driver to offer transport services from MITU offices in Mwanza city to rural districts in Mwanza, Geita, Shinyanga and Simiyu regions.
- The vehicle should be a station Wagon with a capacity of carrying a minimum of eight passengers excluding the driver.
- The vehicle should be fully insured by a comprehensive insurance policy.
- The vehicle should be in good condition with all necessary safety measures (seat belts, first aid kit etc) in place.
- The bidder will meet cost for vehicle repair and periodic scheduled vehicle maintenance.
- The bidder will ensure the vehicle is clean and comfortable for the passengers.
MITU will cover costs for fuel following its strict fuel control system, and road and ferry tolls where applicable.
Terms and Conditions
We request bidders to indicate the following in their bids:
- Monthly rental fees under these conditions specified.
- Number of cars available and when cars will be made available.
How to apply
Interested bidders should submit their expression of interest with the title “Supply of vehicle and a driver” preferably through email address recruitment@mitu.or.tz or the address below:
The Mwanza Intervention Trials Unit
P.O Box 11936
Mwanza
The application should be submitted before 22nd January 2023.

About the course
This is a two-week, practical research methods course suitable for health researchers, social scientists and students undertaking professional training (MSc, MMed, MPH, and PhD). Facilitators for this course are highly experienced researchers from the local and international research institutions. The course consists of eight introductory modules on Epidemiology, Medical statistics, Bioethics, Field methods and Implementation Research, Laboratory methods and evaluation, Qualitative research methods, Clinical trials and Scientific critical reading and referencing.
For full course details and how to apply, please download the printable course advert using the download button below.
Application deadline
Applications close on Friday, 13th January 2023 at 05:00 pm East Africa Time.
Share with your networks
If you are interested in sharing this research methods course within your own networks, please download the printable advert below.
If you would like more details about the program, please contact rmcourse@mitu.or.tz

Mwanza Intervention Trials Unit (MITU) has launched a new trial of human papilloma virus (HPV) vaccine (“Add-Vacc Trial”). HPV vaccination has been included within the Tanzania national vaccination programme since 2018 and is delivered to girls around 14 years of age to protect them from cervical cancer. Apart from being the main cause of cervical cancer, HPV virus also causes other health problems including anogenital warts and other less common forms of cancer, such as penile cancer, anal cancer and throat cancer.

Staged consent process
Over the course of three years, the Add-Vacc trial will investigate the benefits of adding male vaccination to the existing national programme of female HPV vaccination in Tanzania. A number of countries have begun vaccinating boys in addition to girls – known as gender neutral vaccination. Vaccinating males can protect them from health problems caused by HPV virus infection while at the same time preventing them from passing the virus on to their unvaccinated female partners. This could help reduce the amount of HPV infection and associated health problems in the community.

Country side where the trial will take place
Twenty-six communities will be randomly assigned to receive either the intervention (male HPV vaccination plus female vaccination as part of the ongoing national vaccination programme) or the control condition (only female vaccination as part of the ongoing national vaccination programme). A single dose of male HPV vaccination will be provided to males 14-18 years of age. Two household surveys will be conducted – one before male vaccination is implemented and another one three years later, to assess whether this added vaccination reduces the amount of HPV infection present in the communities. Social science research will be conducted as part of this trial to help the research team to better understand community knowledge and perceptions of HPV vaccination.
Dr. Victoria Ewing who is coordinating the implementation of this trial said, “This trial is important because giving a single dose of HPV vaccine to males might provide a simple and cost-effective way to reduce the amount of HPV infection in the community. We are excited to be the first to conduct a trial of HPV vaccination in boys in sub-Saharan Africa.”
This trial is led by Prof. Debby Watson-Jones and Dr. Kathy Baisley from the London School of Hygiene & Tropical Medicine, and John Changalucha from MITU.




ABOUT THE PROGRAM
Supported by the Fogarty International Center at the National Institutes of Health, this research training program is designed to increase the number of clinical investigators in Mwanza with rigorous training in patient-oriented HIV research. Applicants interested in mentored research training in the following HIV-related research topics are encouraged to apply:
- HIV prevention and vaccine research
- Implementation of HIV testing and treatment
- Women’s health and HIV
- HIV and co-infections
- HIV and cardiovascular disease.
Participants in the program may be sponsored to enroll in the MSc in Epidemiology and Biostatistics or PhD program at the Catholic University of Health and Allied Sciences (CUHAS).
APPLICATION REQUIREMENTS
The following documents are required to apply for the Fogarty Research Training Program:
- One-page summary of your education and experience to date, your career goals, and a brief summary of your proposed research project
- Curriculum Vitae
- Letter of support from your mentor
APPLICATION PROCESS
For full details on the application requirements and application process, please visit the Application Portal webpage using the link below.
APPLICATION DEADLINE
Applications close on 1 May 2022 at 11:59 EAT.
SHARE WITH YOUR NETWORK
If you are interested in sharing this research training program within your own networks, please download the printable poster and flyer below.
If you would like more details about the program, please contact lir2020@med.cornell.edu
Source:
NIH Director’s Blog

Credit: Adapted from Pfizer, Dec. 8, 2021
There’s been great concern about the new Omicron variant of SARS-CoV-2, the coronavirus that causes COVID-19. A major reason is Omicron has accumulated over 50 mutations, including about 30 in the spike protein, the part of the coronavirus that mRNA vaccines teach our immune systems to attack. All of these genetic changes raise the possibility that Omicron could cause breakthrough infections in people who’ve already received a Pfizer or Moderna mRNA vaccine.
So, what does the science show? The first data to emerge present somewhat encouraging results. While our existing mRNA vaccines still offer some protection against Omicron, there appears to be a significant decline in neutralizing antibodies against this variant in people who have received two shots of an mRNA vaccine.
However, initial results of studies conducted both in the lab and in the real world show that people who get a booster shot, or third dose of vaccine, may be better protected. Though these data are preliminary, they suggest that getting a booster will help protect people already vaccinated from breakthrough or possible severe infections with Omicron during the winter months.
Though Omicron was discovered in South Africa only last month, researchers have been working around the clock to learn more about this variant. Last week brought the first wave of scientific data on Omicron, including interesting work from a research team led by Alex Sigal, Africa Health Research Institute, Durban, South Africa [1].
In lab studies working with live Omicron virus, the researchers showed that this variant still relies on the ACE2 receptor to infect human lung cells. That’s really good news. It means that the therapeutic tools already developed, including vaccines, should generally remain useful for combatting this new variant.
Sigal and colleagues also tested the ability of antibodies in the plasma from 12 fully vaccinated individuals to neutralize Omicron. Six of the individuals had no history of COVID-19. The other six had been infected with the original variant in the first wave of infections in South Africa.
As expected, the samples showed very strong neutralization against the original SARS-CoV-2 variant. However, antibodies from people who’d been previously vaccinated with the two-dose Pfizer vaccine took a significant hit against Omicron, showing about a 40-fold decline in neutralizing ability.
This escape from immunity wasn’t complete. Indeed, blood samples from five individuals showed relatively good antibody levels against Omicron. All five had previously been infected with SARS-CoV-2 in addition to being vaccinated. These findings add to evidence on the value of full vaccination for protecting against reinfections in people who’ve had COVID-19 previously.
Also of great interest were the first results of the Pfizer study, which the company made available in a news release [2]. Pfizer researchers also conducted laboratory studies to test the neutralizing ability of blood samples from 19 individuals one month after a second shot compared to 20 others one month after a booster shot.
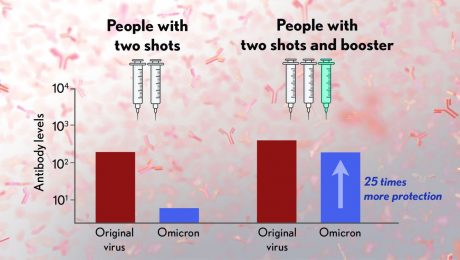
These studies showed that the neutralizing ability of samples from those who’d received two shots had a more than 25-fold decline relative to the original virus. Together with the South Africa data, it suggests that the two-dose series may not be enough to protect against breakthrough infections with the Omicron variant.
In much more encouraging news, their studies went on to show that a booster dose of the Pfizer vaccine raised antibody levels against Omicron to a level comparable to the two-dose regimen against the original variant (as shown in the figure above). While efforts already are underway to develop an Omicron-specific COVID-19 vaccine, these findings suggest that it’s already possible to get good protection against this new variant by getting a booster shot.
Very recently, real-world data from the United Kingdom, where Omicron cases are rising rapidly, are providing additional evidence for how boosters can help. In a preprint [3], Andrews et. al showed the effectiveness of two shots of Pfizer mRNA vaccine trended down after four months to about 40 percent. That’s not great, but note that 40 percent is far better than zero. So, clearly there is some protection provided.
Graph showing Pfizer booster is about 80% effective after 2 weeks against Omicron

Credit: Andrews N, et al., KHub.net 2021
Most impressively (as shown in the figure from Andrews N, et al.) a booster substantially raised that vaccine effectiveness to about 80 percent. That’s not quite as high as for Delta, but certainly an encouraging result. Once again, these data show that boosting the immune system after a pause produces enhanced immunity against new viral variants, even though the booster was designed from the original virus. Your immune system is awfully clever. You get both quantitative and qualitative benefits.
It’s also worth noting that the Omicron variant mostly doesn’t have mutations in portions of its genome that are the targets of other aspects of vaccine-induced immunity, including T cells. These cells are part of the body’s second line of defense and are generally harder for viruses to escape. While T cells can’t prevent infection, they help protect against more severe illness and death.
It’s important to note that scientists around the world are also closely monitoring Omicron’s severity While this variant appears to be highly transmissible, and it is still early for rigorous conclusions, the initial research indicates this variant may actually produce milder illness than Delta, which is currently the dominant strain in the United States.
But there’s still a tremendous amount of research to be done that could change how we view Omicron. This research will take time and patience.
What won’t change, though, is that vaccines are the best way to protect yourself and others against COVID-19. (And these recent data provide an even-stronger reason to get a booster now if you are eligible.) Wearing a mask, especially in public indoor settings, offers good protection against the spread of all SARS-CoV-2 variants. If you’ve got symptoms or think you may have been exposed, get tested and stay home if you get a positive result. As we await more answers, it’s as important as ever to use all the tools available to keep yourself, your loved ones, and your community happy and healthy this holiday season.
References:
[1] SARS-CoV-2 Omicron has extensive but incomplete escape of Pfizer BNT162b2 elicited neutralization and requires ACE2 for infection . Sandile C, et al. Sandile C, et al. medRxiv preprint. December 9, 2021. [2] Pfizer and BioNTech provide update on Omicron variant . Pfizer. December 8, 2021. [3] Effectiveness of COVID-19 vaccines against the Omicron (B.1.1.529) variant of concern . Andrews N, et al. KHub.net preprint. December 10, 2021.Links:
COVID-19 Research (NIH)
Sigal Lab (Africa Health Research Institute, Durban, South Africa)

The COVID-19 pandemic has brought a radical shift in the work environment, resulting in increased number of people around the world working from home or other locations away from their official work space.

 MITU did not lag behind in protecting its staff from the spread of the pandemic and allowed a vast majority of staff to work from home. Fortunately, the technology and the available ICT infrastructure at the Unit supported this move and helped the remote working staff remain connected and productive.
MITU did not lag behind in protecting its staff from the spread of the pandemic and allowed a vast majority of staff to work from home. Fortunately, the technology and the available ICT infrastructure at the Unit supported this move and helped the remote working staff remain connected and productive.
MITU is connected through fibre internet – a broadband connection that uses fibre-optic technology to provide fastest speeds data over greater distances without major lag time. With such infrastructure in place, staff are connecting to box drive (a cloud storage service) with ease enabling them to share documents remotely from anywhere anytime. With laptop computers, USB speakerphones, headphones and the internet, staff have been able to stay connected and maintain workflow continuity and conduct live meetings and presentations using Zoom – a video conferencing web platform.
With a virtual private network (VPN) connection – a secure tunnelling technology, staff have been able to securely access the Unit’s local resources from the unsecure public internet by connecting from outside MITU. Remote support tools have also enabled the IT technicians gain access to the remote laptops used by staff working from home and provide technical support in cases of need.
The COVID-19 pandemic has unleashed the new ways in which ICT could support remote working arrangements and allow organizations to remain productive during these challenging times.